Palladium-Catalyzed Ring-Closing Reaction via C–N Bond Metathesis for Rapid Construction of Saturated N-Heterocycles | Journal of the American Chemical Society
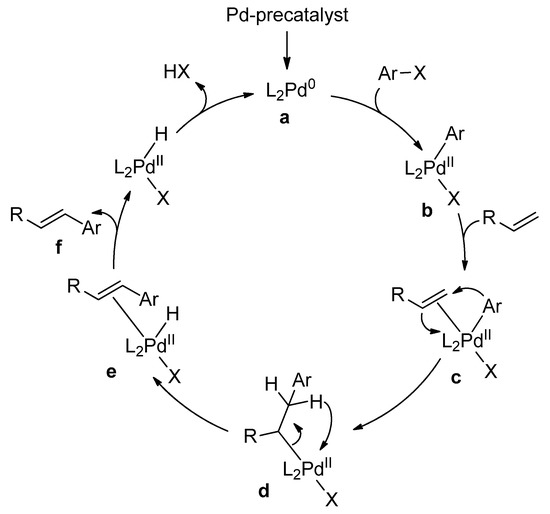
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond

Palladium-Catalyzed Regioselective and Stereospecific Ring-Opening Cross-Coupling of Aziridines: Experimental and Computational Studies | Accounts of Chemical Research
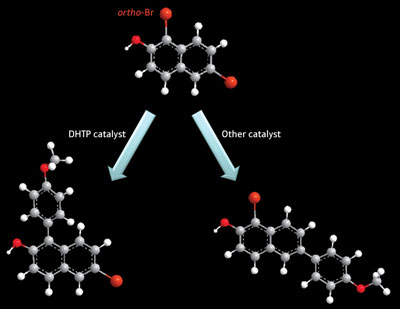
Palladium catalysts containing unique molecular ligands couple aromatic rings together in surprising ways

Palladium-catalyst development for the intermolecular carbonylative... | Download Scientific Diagram

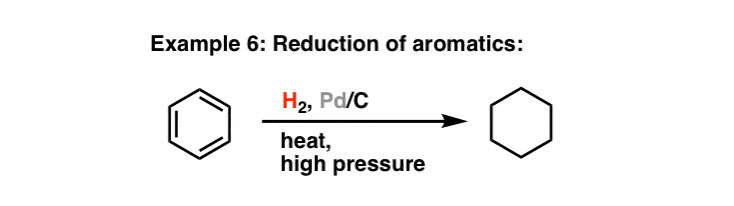
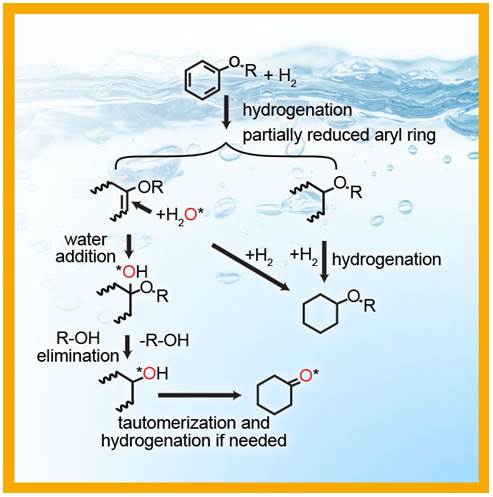
The application of a supported palladium catalyst for the hydrogenation of aromatic nitriles - ScienceDirect
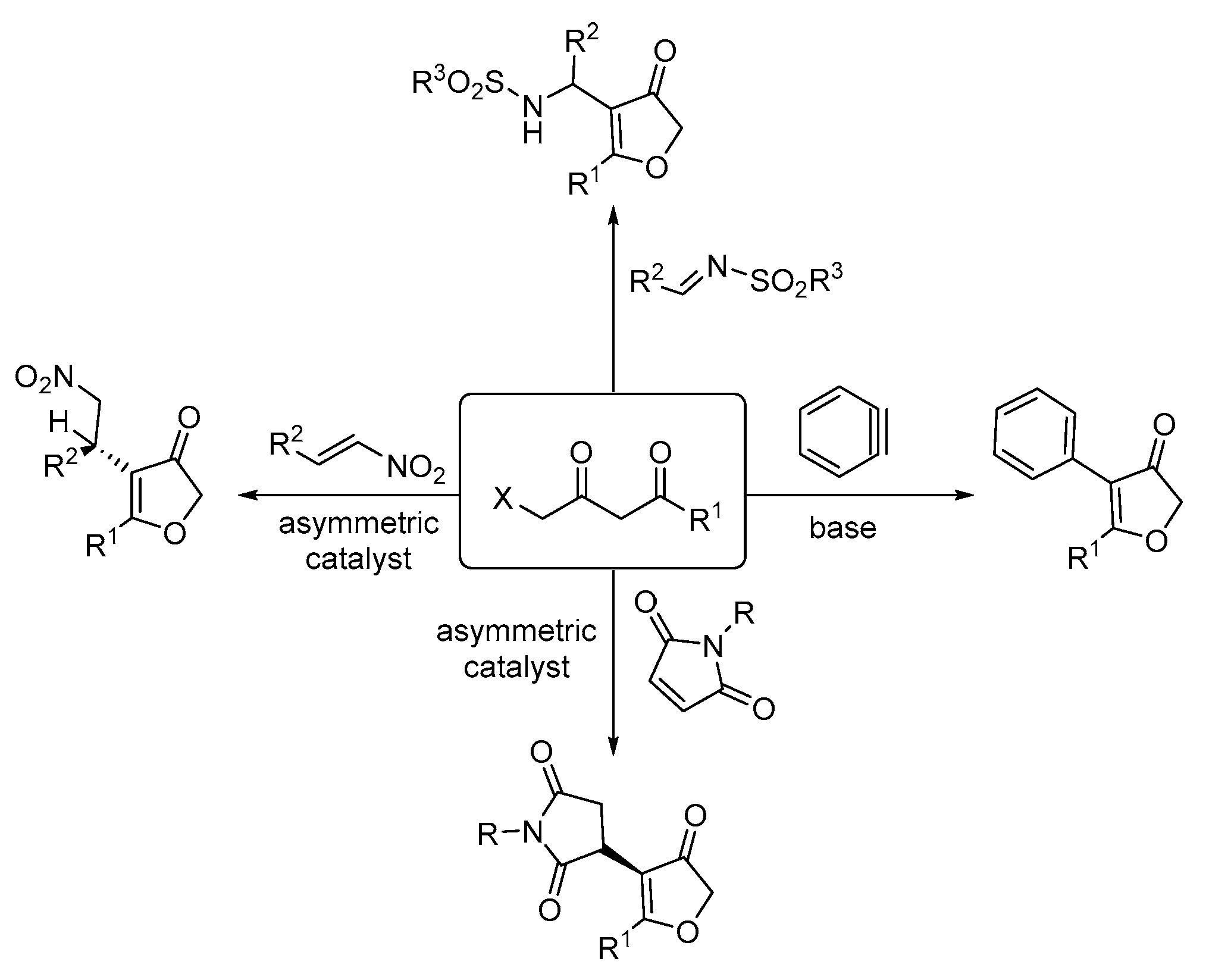
Organics | Free Full-Text | Palladium Catalyzed Ring-Opening of Diazabicylic Olefins with 4-Halo-1,3-Dicarbonyl Compounds: Accessing 3(2H)-Furanone-Appended Cyclopentenes

Pd-Catalyzed Ring-Closing/Ring-Opening Cross Coupling Reactions: Enantioselective Diarylation of Unactivated Olefins | ACS Catalysis

Palladium-catalyzed oxidative arene C–H alkenylation reactions involving olefins: Trends in Chemistry
![PDF] A hydroquinone based palladium catalyst for room temperature nitro reduction in water | Semantic Scholar PDF] A hydroquinone based palladium catalyst for room temperature nitro reduction in water | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7f50878b12ee80f4266e9a654281d70727457521/2-Table2-1.png)
PDF] A hydroquinone based palladium catalyst for room temperature nitro reduction in water | Semantic Scholar

General representation of the arylic C-X bond activation by a palladium... | Download Scientific Diagram

Ring Or Ball Type Nickel Catalyst Ball, Cube, Ribs,, For Industrial, Grade Standard: Analytical Grade

Aerobic Heterogeneous Palladium-Catalyzed Oxidative Allenic C−H Arylation: Benzoquinone as a Direct Redox Mediator between O2 and Pd | CCS Chemistry