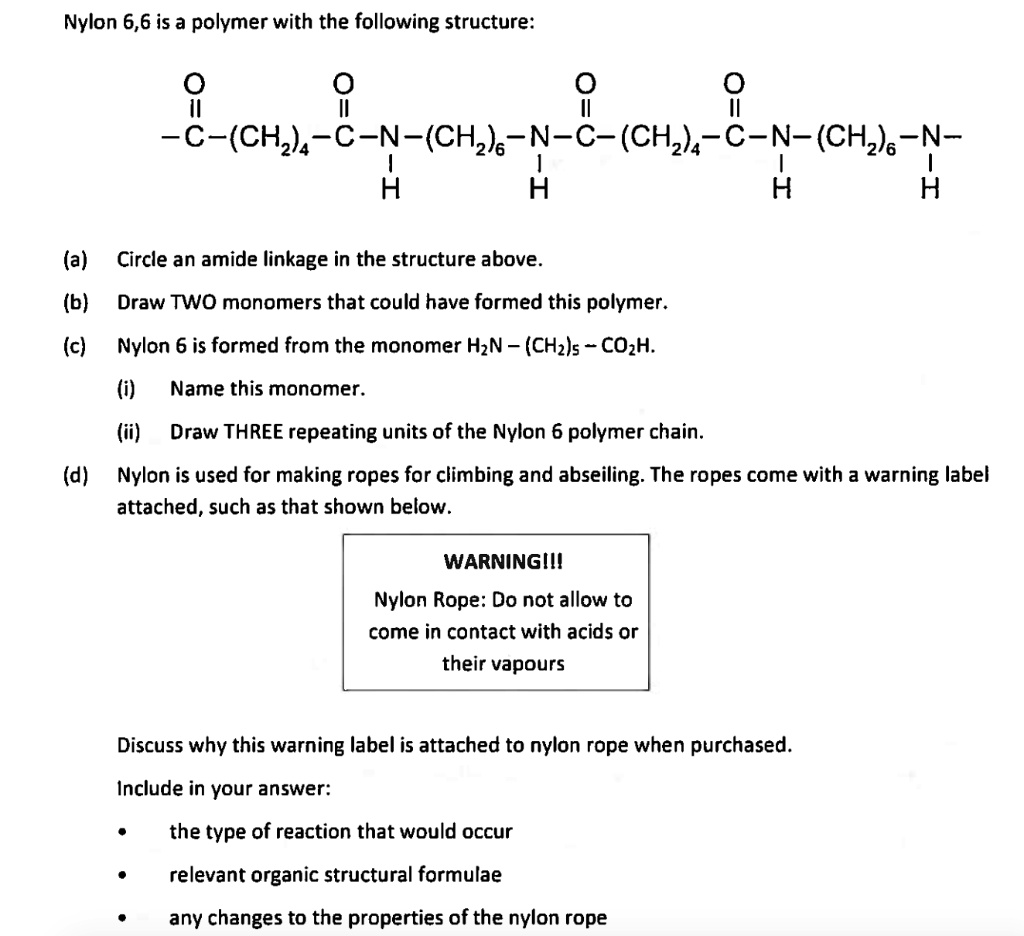
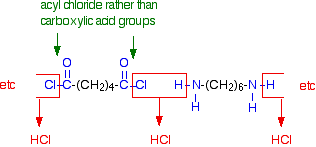
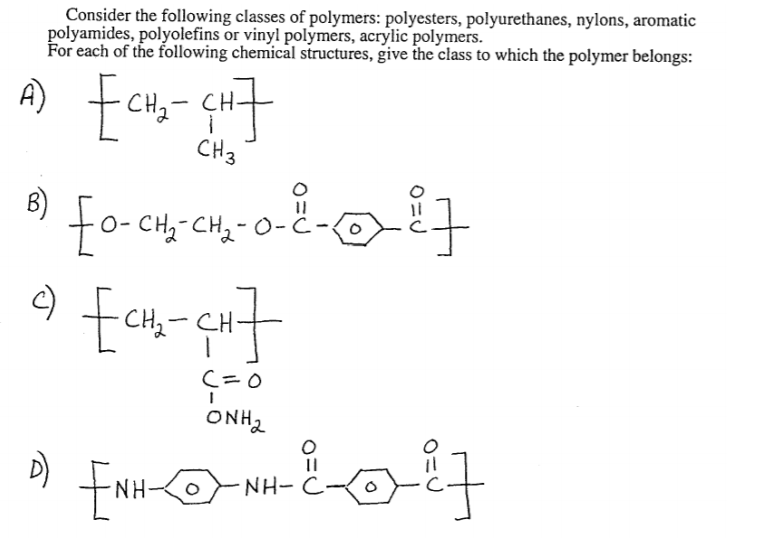
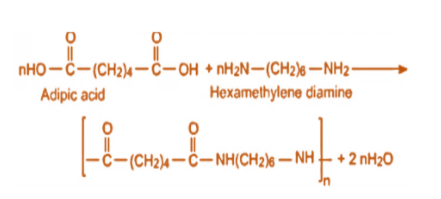
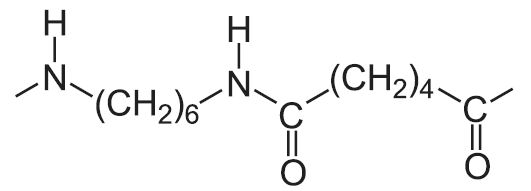
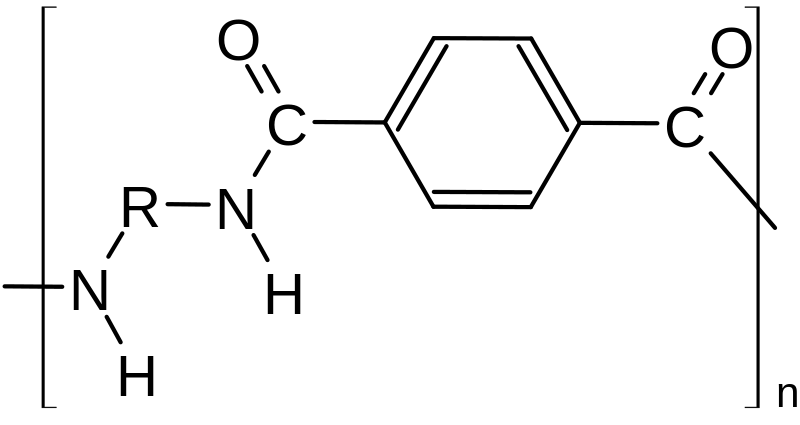
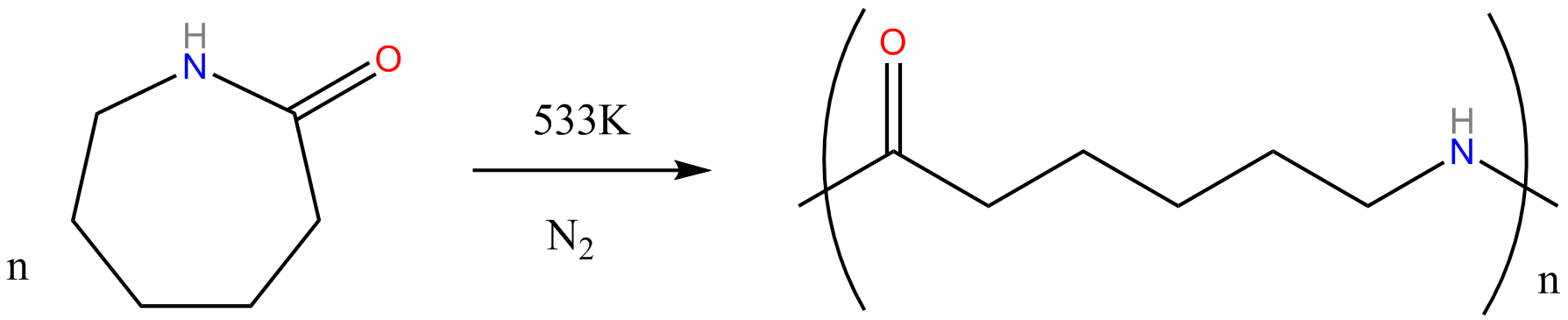
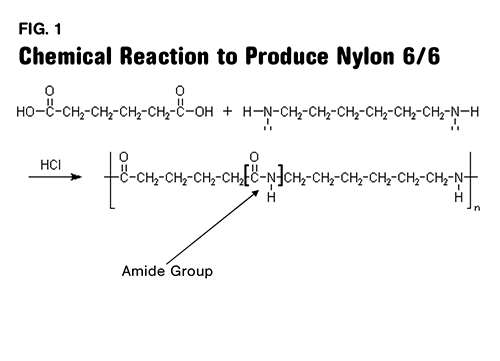
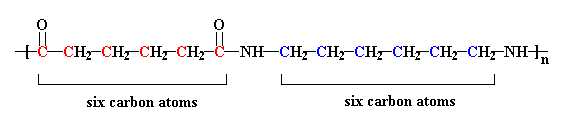
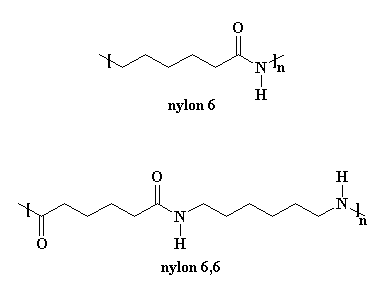
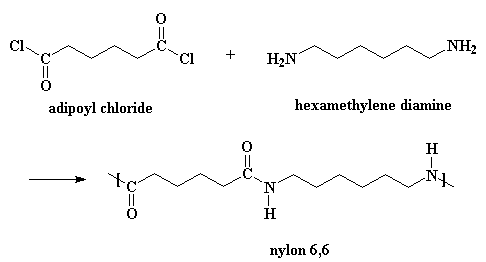
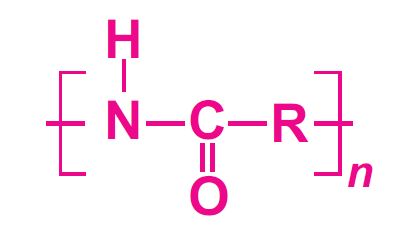Polyamides are polymers that contain many amide groups. These groups form when a carboxylic acid reacts with an amine. - ppt download

Polymer repeating units: (a) Nylon 6 (polycaprolactame, the homopolymer... | Download Scientific Diagram

Draw the structure of Nylon-4,4 (draw 3 repeating units). What monomers would be needed for the formation of Nylon-4,4 (provide the structures)? | Homework.Study.com