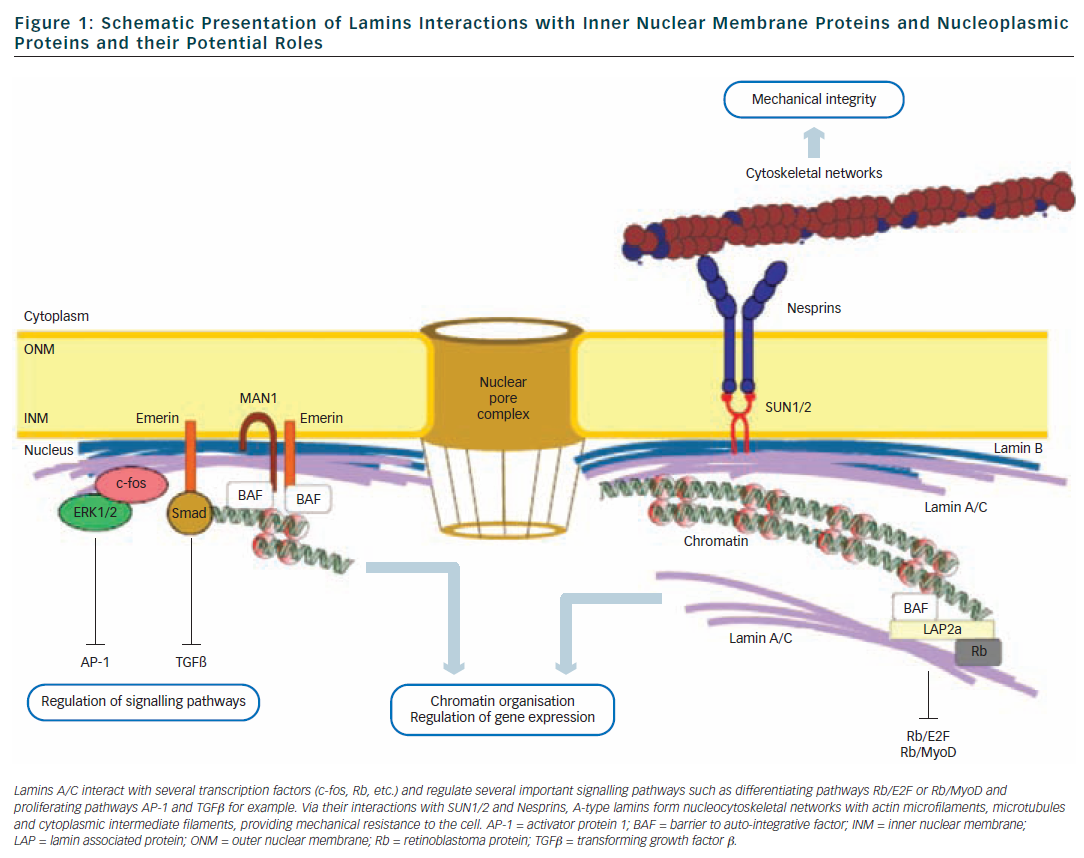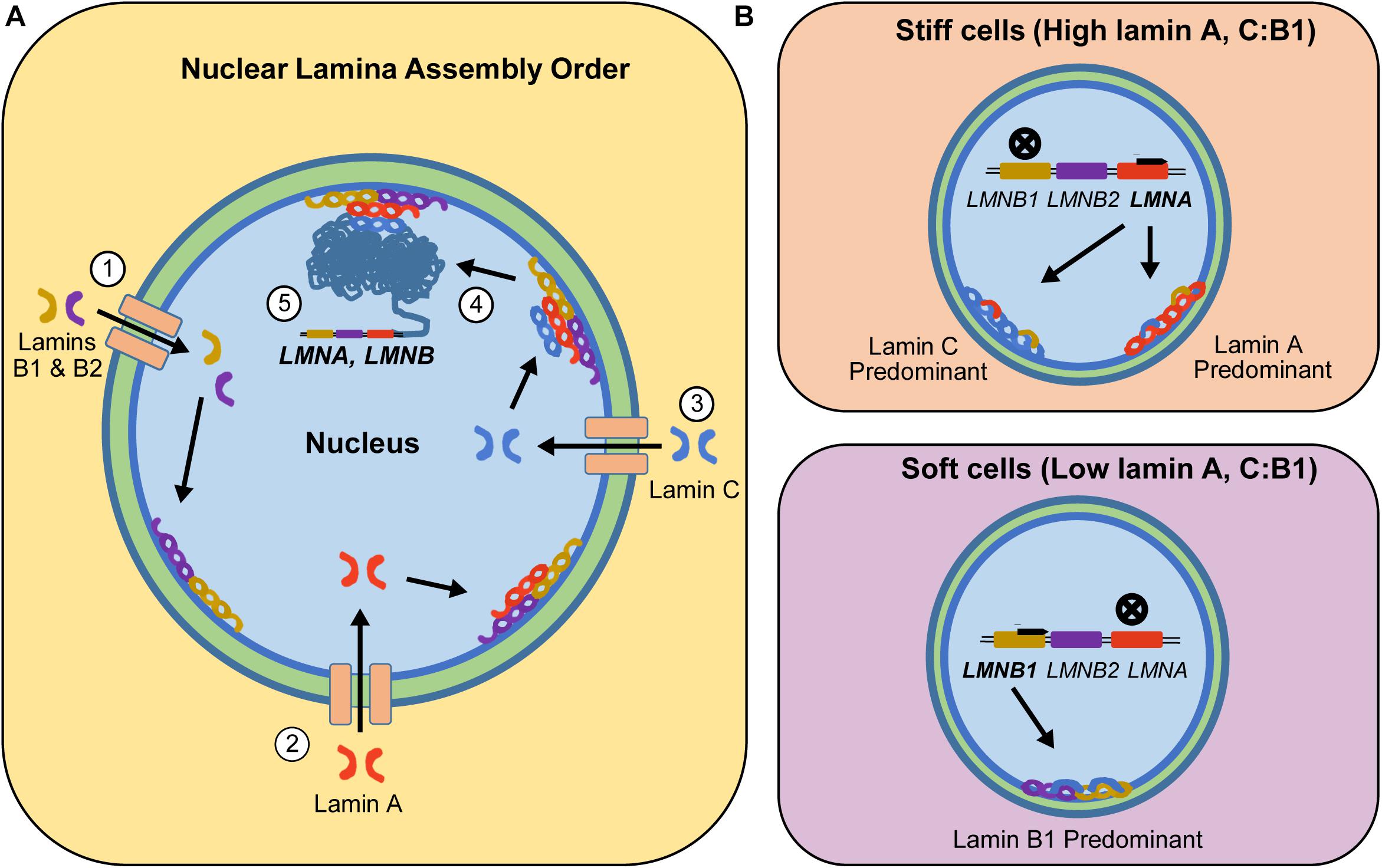
Frontiers | Diversity of Nuclear Lamin A/C Action as a Key to Tissue-Specific Regulation of Cellular Identity in Health and Disease

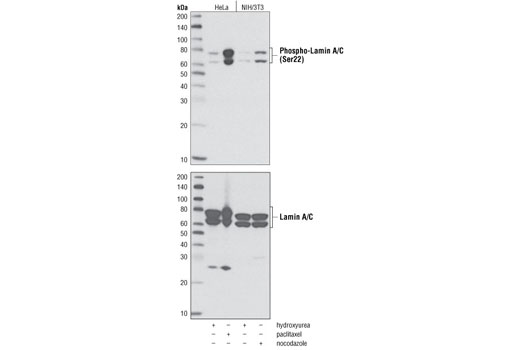
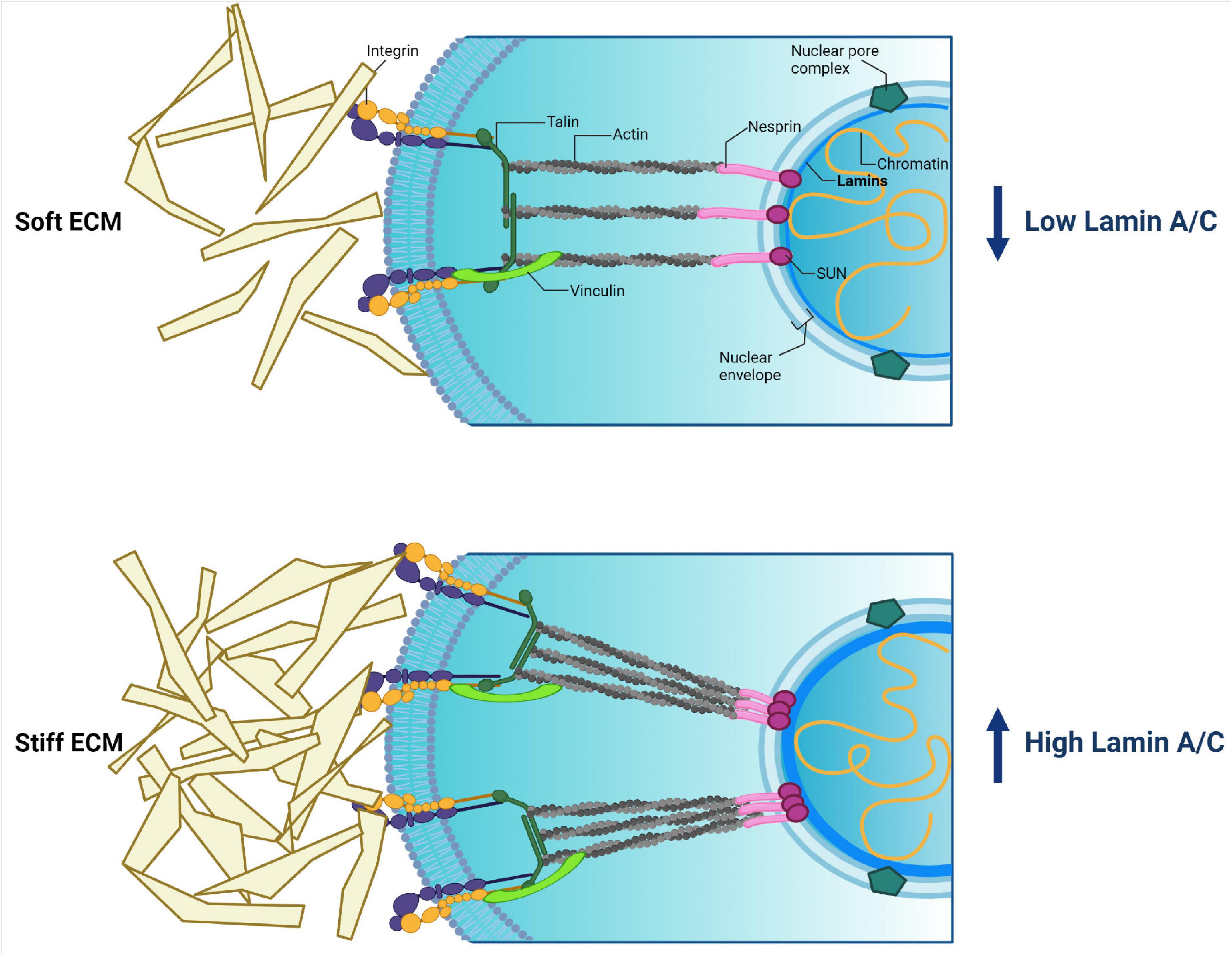
Matrix Elasticity Regulates Lamin-A,C Phosphorylation and Turnover with Feedback to Actomyosin: Current Biology
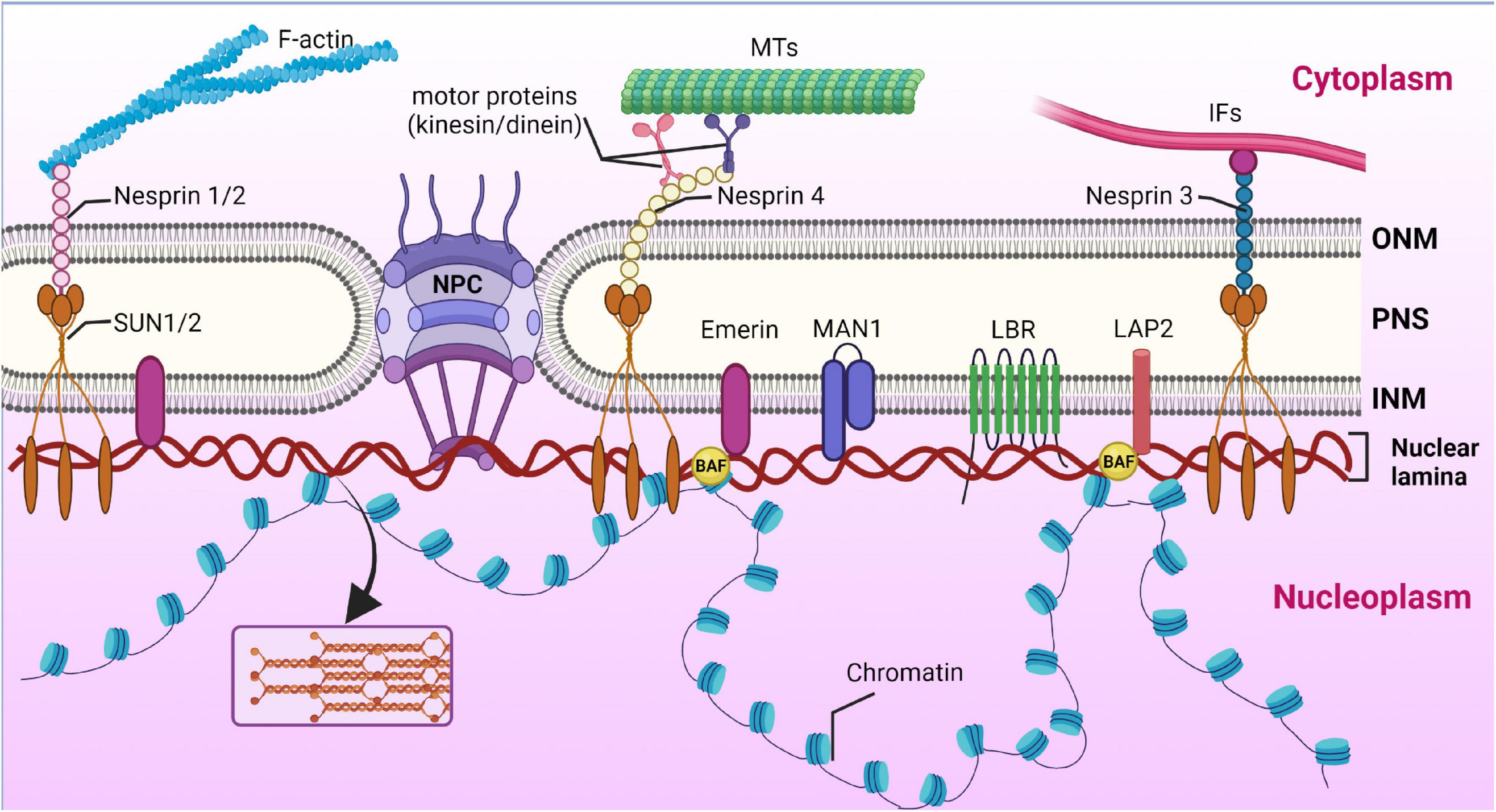
Frontiers | Diversity of Nuclear Lamin A/C Action as a Key to Tissue-Specific Regulation of Cellular Identity in Health and Disease

LBR and Lamin A/C Sequentially Tether Peripheral Heterochromatin and Inversely Regulate Differentiation - ScienceDirect

Missense Mutations in the Rod Domain of the Lamin A/C Gene as Causes of Dilated Cardiomyopathy and Conduction-System Disease | NEJM
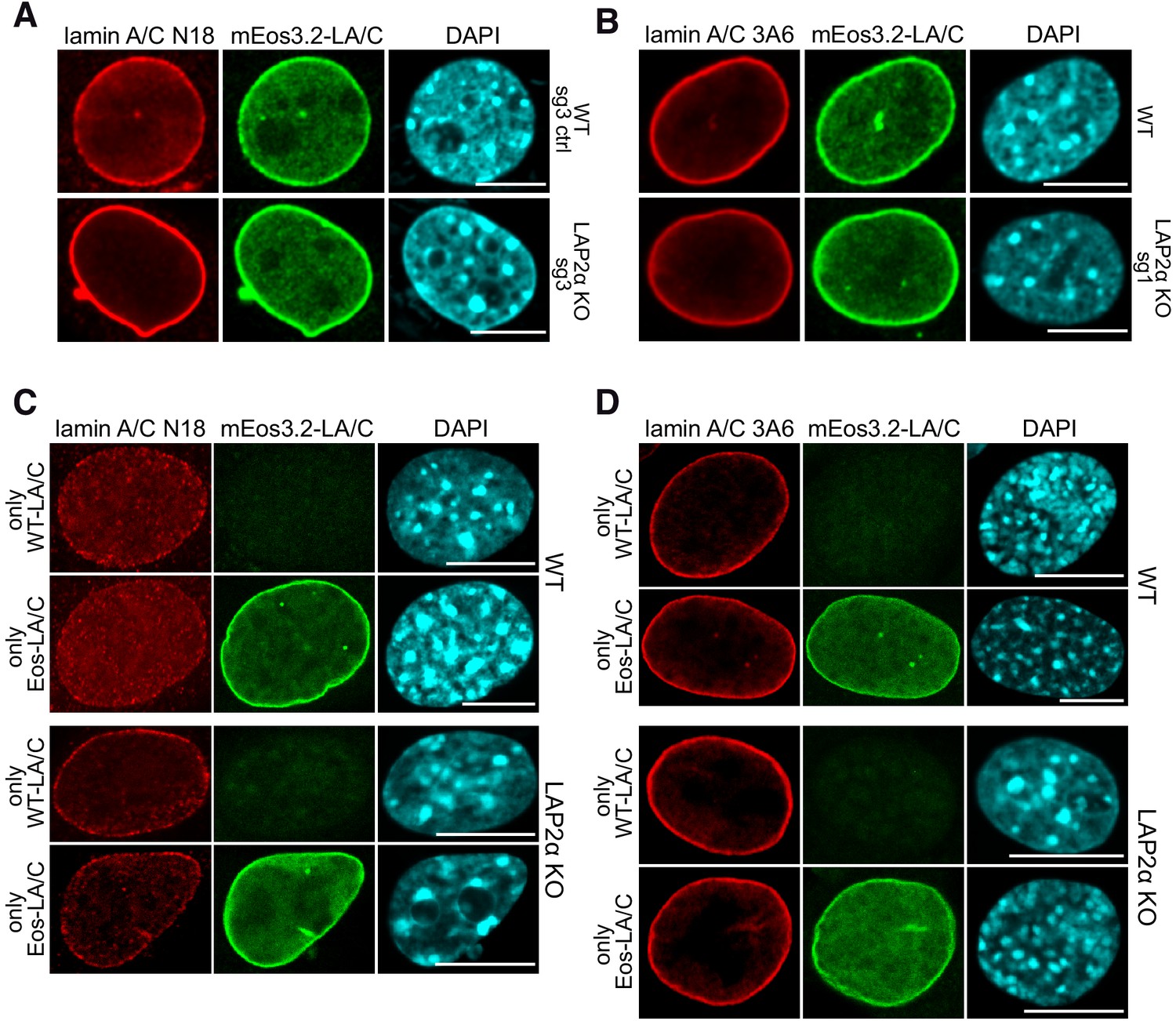
LAP2alpha maintains a mobile and low assembly state of A-type lamins in the nuclear interior | eLife

Tyrosine phosphorylation of lamin A by Src promotes disassembly of nuclear lamina in interphase | Life Science Alliance
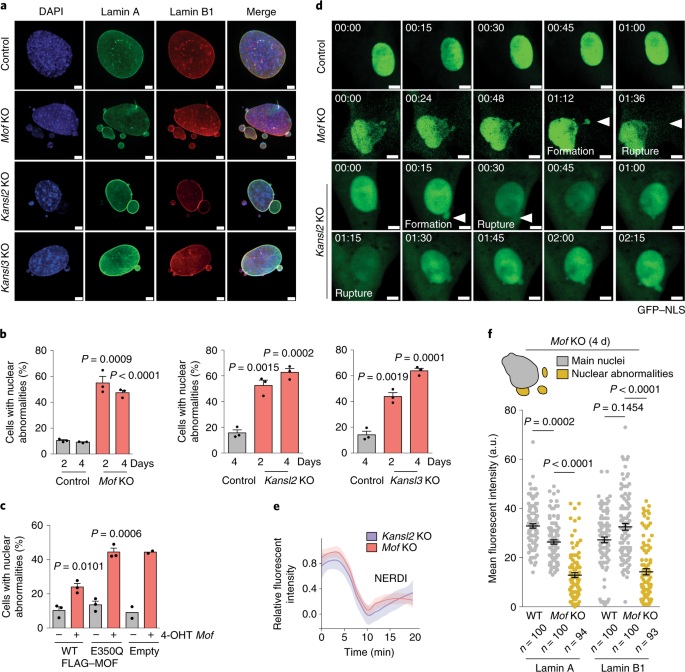
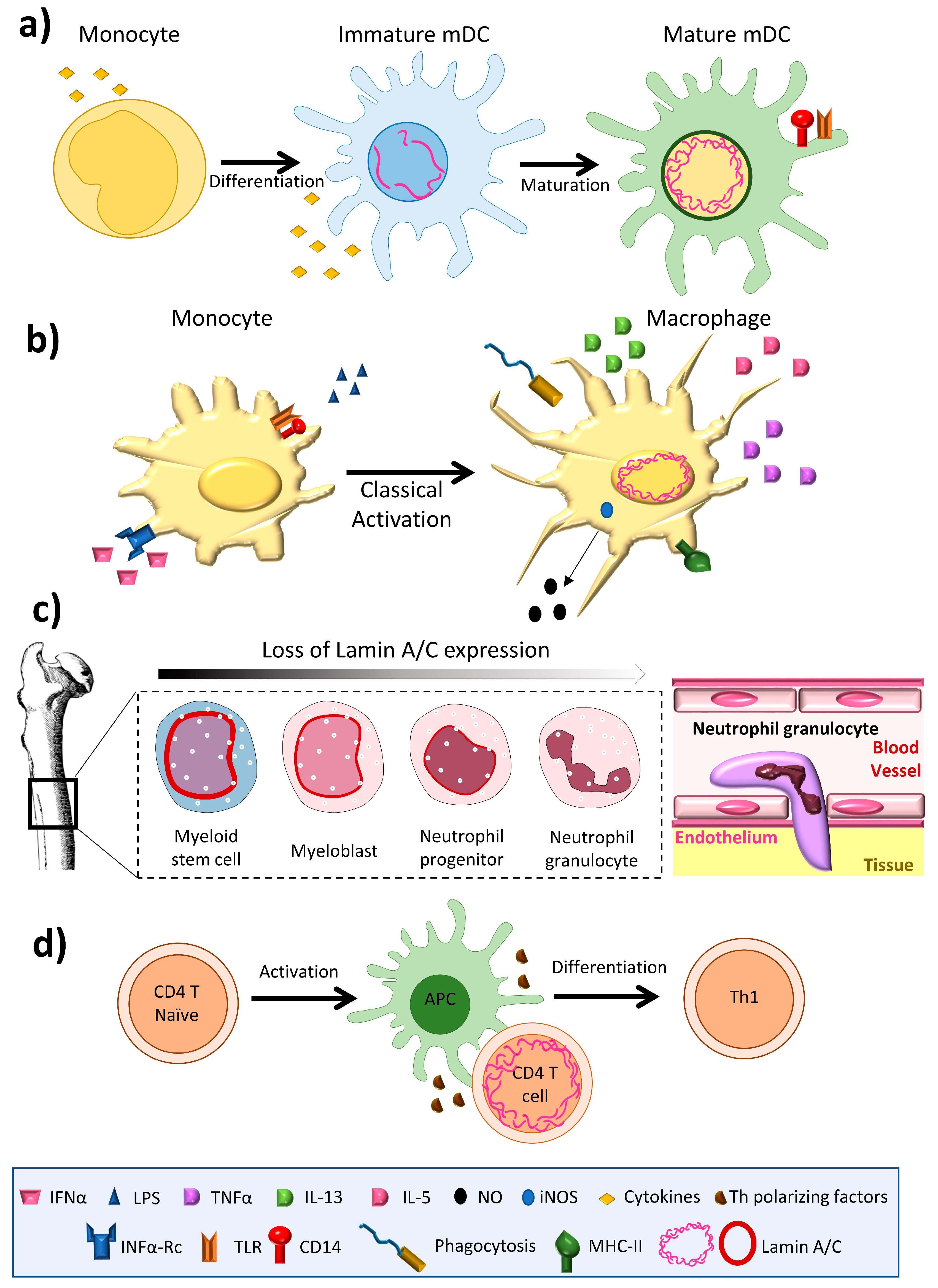
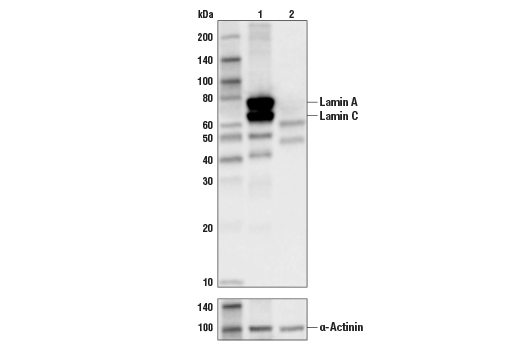
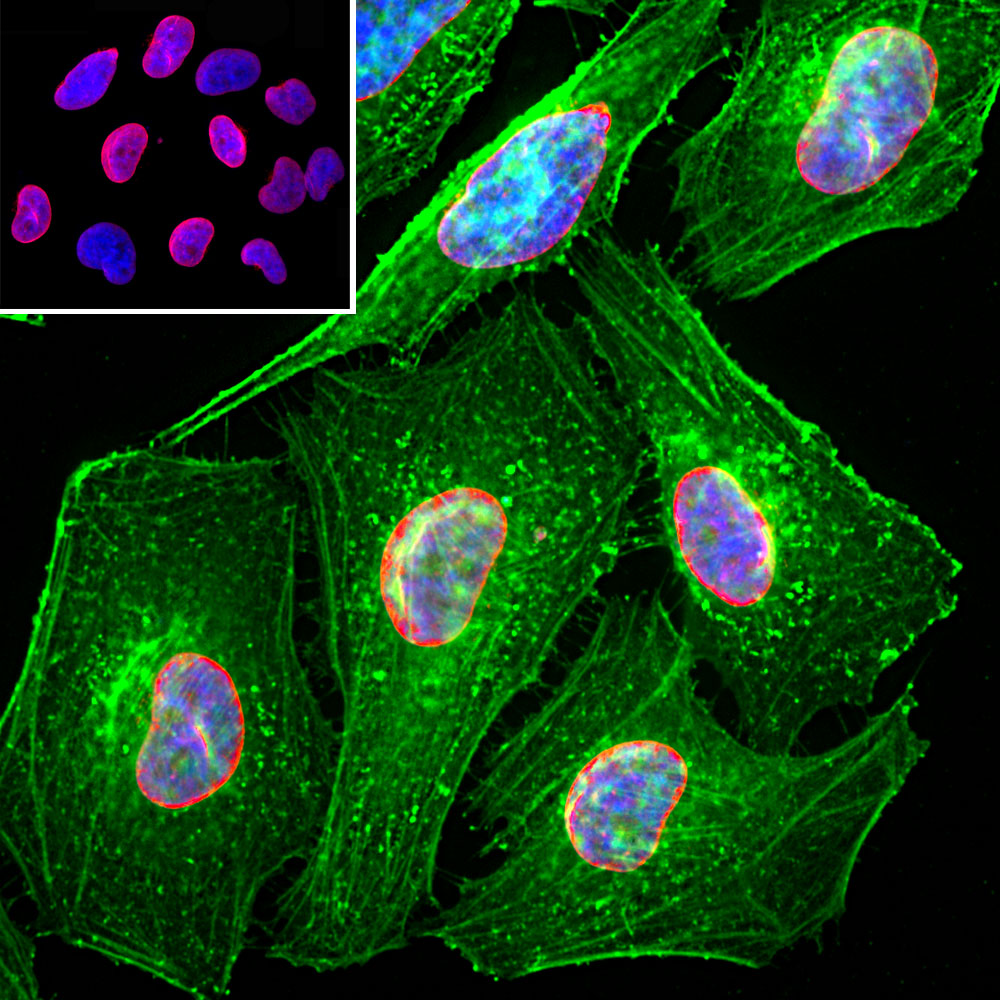
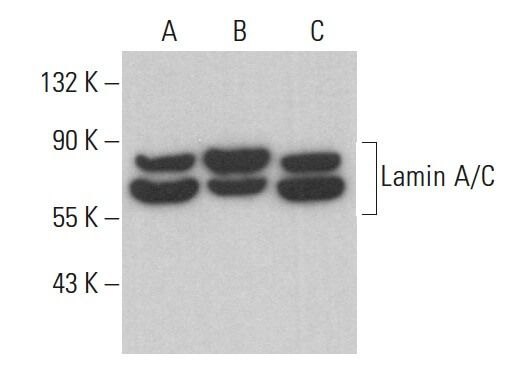

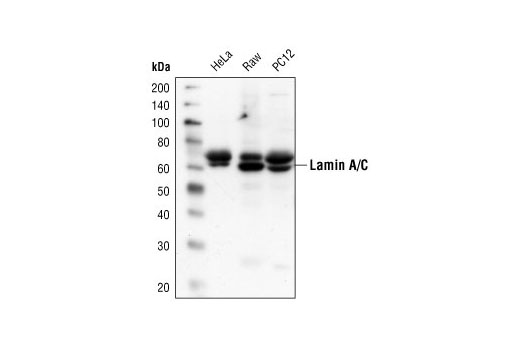



![Lamin A+C Monoclonal Antibody [JOL2] - ImmuQuest Lamin A+C Monoclonal Antibody [JOL2] - ImmuQuest](https://immuquest.com/cdn/shop/products/IQ332-IF.jpg?v=1662642780)